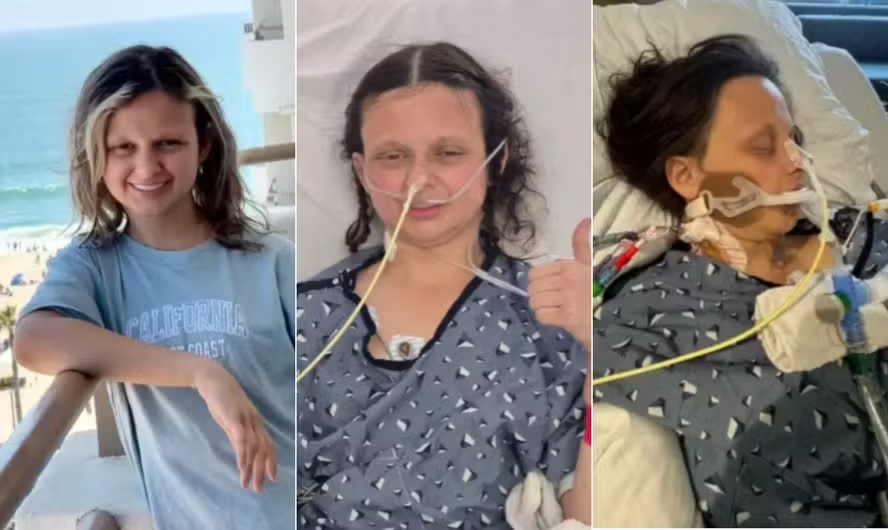
In a major medical breakthrough, researchers say a new gene therapy trial has delivered results that could change what was thought possible for Huntington’s disease, a genetic brain disorder that has long been considered untreatable. The trial, led by scientists at University College London in collaboration with the biotech company uniQure, treated patients with the therapy called AMT-130 using a one-time brain surgery procedure.
The treatment involves injecting modified DNA into parts of the brain to block the production of the toxic huntingtin protein, the cause of Huntington’s disease. For patients receiving high doses of the therapy, data shows the disease’s progression was slowed by about 75 percent after three years. The improvements were seen in motor function, thinking ability, and daily life tasks. Researchers also found markers of brain cell damage were much lower in treated patients compared to those who did not get the therapy.
Doctors say this may be the first therapy that does more than just ease symptoms—it appears to affect the course of the disease. Huntington’s disease usually appears in mid-life and steadily worsens, causing loss of movement control, mental decline, mood changes, and eventually loss of ability to speak or walk. Before this, treatments could only address symptoms like involuntary movements or depression; nothing had been shown to slow how fast the disease moves forward.
The trial included 29 patients across the UK and the U.S., though only a subgroup got the highest doses. Those in that subgroup saw the most dramatic slowing. Others had smaller benefits, depending on dose. Because the surgery is complex and long (12-20 hours), it’s not an easy fix. The procedure requires careful delivery of the therapy into two separate regions of the brain.
Researchers caution that the results are early. Full data is still being reviewed, and long-term effects are not yet known. They also say the therapy is expensive and difficult to administer, which may limit how quickly it becomes widely available. There are also risks that come with brain surgery, and not all patients may be good candidates.
Still, patients and their families are speaking of renewed hope. Some said that just having something that appears to slow Huntington’s feels like a turning point. For many years, people living with the disease have been waiting for a treatment that does more than manage symptoms. This development brings forward the possibility of earlier diagnosis and intervention—maybe even before symptoms start.
With this success, uniQure plans to apply for regulatory approval in the U.S., likely in early 2026. If approved, AMT-130 could become the first licensed treatment to slow disease progression rather than just help with symptoms.
For now, though, experts say there’s a lot of work to be done: more trials, more patients, and more data. Accessibility, cost, patient eligibility, and long-term safety are all questions that remain. But for a condition that has caused so much suffering, many see this as a moment of breakthrough.